Clinical Research
Clinical Research
Patanjali Research Foundation's Clinical Research division focuses on developing astute planning with a commitment to making a difference in the lives of patients by unleashing a complex health landscape of natural and traditional assets through sustainable solutions by translating scientific discoveries from the laboratory (bench-side) to the clinic (bed-side). We are determined to develop evidence-based herbal medicines using a mixture of research, clinical experience, and patient preferences through the clinical trial process.
The Clinical research team consists of an interdisciplinary collaboration of doctors and experts from the fields of allopathy and ayurveda working in harmony, in determining and documenting the safety effectiveness of vivid authentic, classical and patent ayurveda-naturopathy treatment regimens and therapies, intended for human use.
As an organization, we are aiming to supplement our conventional, anecdotal reasoning with high-quality research (pre-clinical studies), observational studies, public health studies, and, finally, the top randomized controlled trial to achieve a healthier world for all.
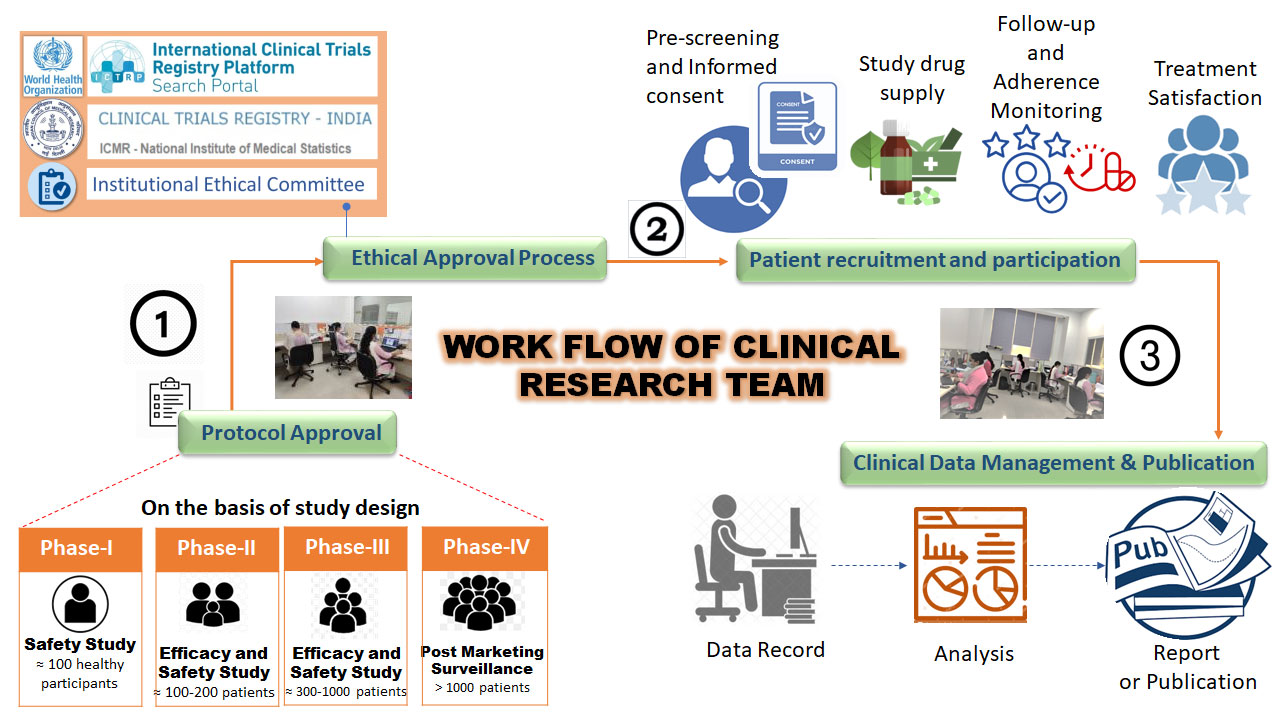
Patanjali Research Foundation's biomedical research is approved by the Institutional Ethical Committee (IEC) and registered with CTRI (Clinical trial registry of India). Patanjali's clinical research team adheres to a code of conduct based on three key ethical principles: respect for persons, beneficence, and justice.
With our dedicated scientific clinical research team at Patanjali
Research Foundation, we leverage to create a better world in which
“Better health outcomes can be provided to all”.